Cartilage rehydration: The sliding-induced hydrodynamic triggering mechanism
Carmine Putignano, David Burris, Axel Moore, Daniele Dini
In this study we have provided an innovative theoretical formulation, corroborated by detailed experiments, which for the first time sheds the light on the hydrodynamic origins of rehydration in cartilage tissues.
When loaded, cartilage starts to lose fluid, thus exudating and causing joint space to thin down: in a long term prospective, this may contribute to tissue degradation and to the insurgence of osteoarthritis, which, together with rheumatoid arthritis, costs £10.2 billion in the NHS and healthcare system now, and is expected to reach an estimated £118.6 billion in the next decade.
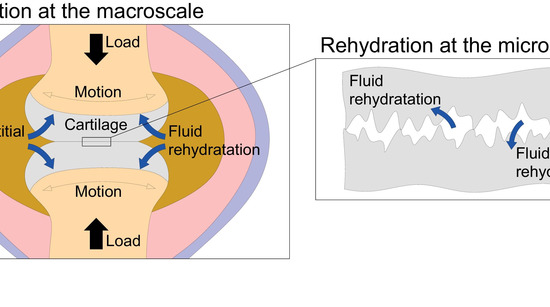
Fortunately, experiments show that, after static loading, cartilage tissues can reverse exudation, recover interstitial fluid and thicken; all of this sets off when joint starts to move. Motion is, therefore, the key to maintain joint hydration and preserve cartilage excellent mechanical properties: however, we have so far missed the theoretical reasons of the motion-induced rehydrative mechanism. This is not only essential to understand the joint physiology, but also to point out a crucial requirement for any material designed to mimic cartilage.
In our work, we present the first original theoretical explanation to the long-standing questions about this fundamental phenomenon, that is, rehydration. By assessing the porous lubrication occurring, at multiple scales, in joints by means of a homogenized treatment of the Reynolds’ Equations, we prove that sliding-induced rehydration occurs because of hydrodynamic reasons. The consistence between numerical predictions and experiments corroborate the robustness of the proposed model.
The insights provided in the work about cartilage rehydration can potentially provide a decisive change to our understanding of cartilage physiology and to our approach to the design of cartilage-mimicking materials.
